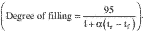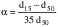HazMat Tool — substance information
Trifluoroacetic acid
HMT May 2026Trifluoroacetic acid
Trifluoroacetic acid (TFA) is a synthetic organofluorine compound with the chemical formula CF3CO2H. It belongs to the subclass of per- and polyfluoroalkyl substances (PFASs) known as ultrashort-chain perfluoroalkyl acids (PFAAs). TFA is not produced biologically or abiotically and is commonly used in organic chemistry for various purposes. It is the most abundant PFAS found in the environment. It is a haloacetic acid, with all three of the acetyl group's hydrogen atoms replaced by fluorine atoms. Wikipedia
| Emergency Response Guide No. 154 |
Hazardous Materials Table
Source: 49 CFR §172 (2026/05)(3) Hazard Class8Corrosive liquids |
(4) Identification number |
(5) Packing Group |
||||||||||||||
(6) Labels |
||||||||||||||||
(7) Special Provisions (§172.102)
|
||||||||||||||||
| A7 | |
|---|---|
| B4 | |
| N3 | |
| N34 | |
| N36 | |
| T10 | |
| TP2 |
(8) Packing Authorizations (§173.***) 
| (8A) Exceptions | None | |
|---|---|---|
| (8B) Non-bulk | 201 | see 49 CFR §173.201 |
| (8C) Bulk | 243 | see 49 CFR §173.243 |
(9) Quantity Limits 
| (9A) Passenger aircraft/rail | 0.5 L |
|---|---|
| (9B) Cargo aircraft only | 2.5 L |
(10) Vessel stowage 
| (10A) Location | |
|---|---|
| (10B) Other |
Segregation chart for load, transport, and storage
This chart shows whether loading, transport, or storage with other hazard classes is permitted, prohibited, or restricted. Based on 49 CFR §177.848.
Note: In this segregation and separation chart are HazMat of hazard class 8 considered depending if there are liquid. Currently HazMat Tool is not able to to check if a HazMat is liquid or not. Therefore it is assumed as liquid. See PHMSA Interpretation #09-0252.
| Hazard class | 8 | 7 | 6.1 | 5.2 | 5.1 | 4.3 | 4.2 | 4.1 | 3 | 2.3B | 2.3A | 2.2 | 2.1 | 1.6 | 1.5 | 1.4 | 1.3 | 1.1/1.2 |
1.1/1.2  | ||||||||||||||||||
1.3  | ||||||||||||||||||
1.4  | ||||||||||||||||||
1.5  | ||||||||||||||||||
1.6  | ||||||||||||||||||
2.1  | ||||||||||||||||||
2.2  | ||||||||||||||||||
2.3A  | ||||||||||||||||||
2.3B  | ||||||||||||||||||
3  | ||||||||||||||||||
4.1  | ||||||||||||||||||
4.2  | ||||||||||||||||||
4.3  | ||||||||||||||||||
5.1  | ||||||||||||||||||
5.2  | ||||||||||||||||||
6.1  | ||||||||||||||||||
7  | ||||||||||||||||||
8  |
| Must not be loaded, transported, or stored together in the same vehicle or facility. | |
| May be loaded, transported, or stored together. | |
| May be loaded, transported, or stored together only with restrictions. |